 Ī calibration equation expresses the quantitative relationship between the response of an analytical technique and the standard concentrations of the target. 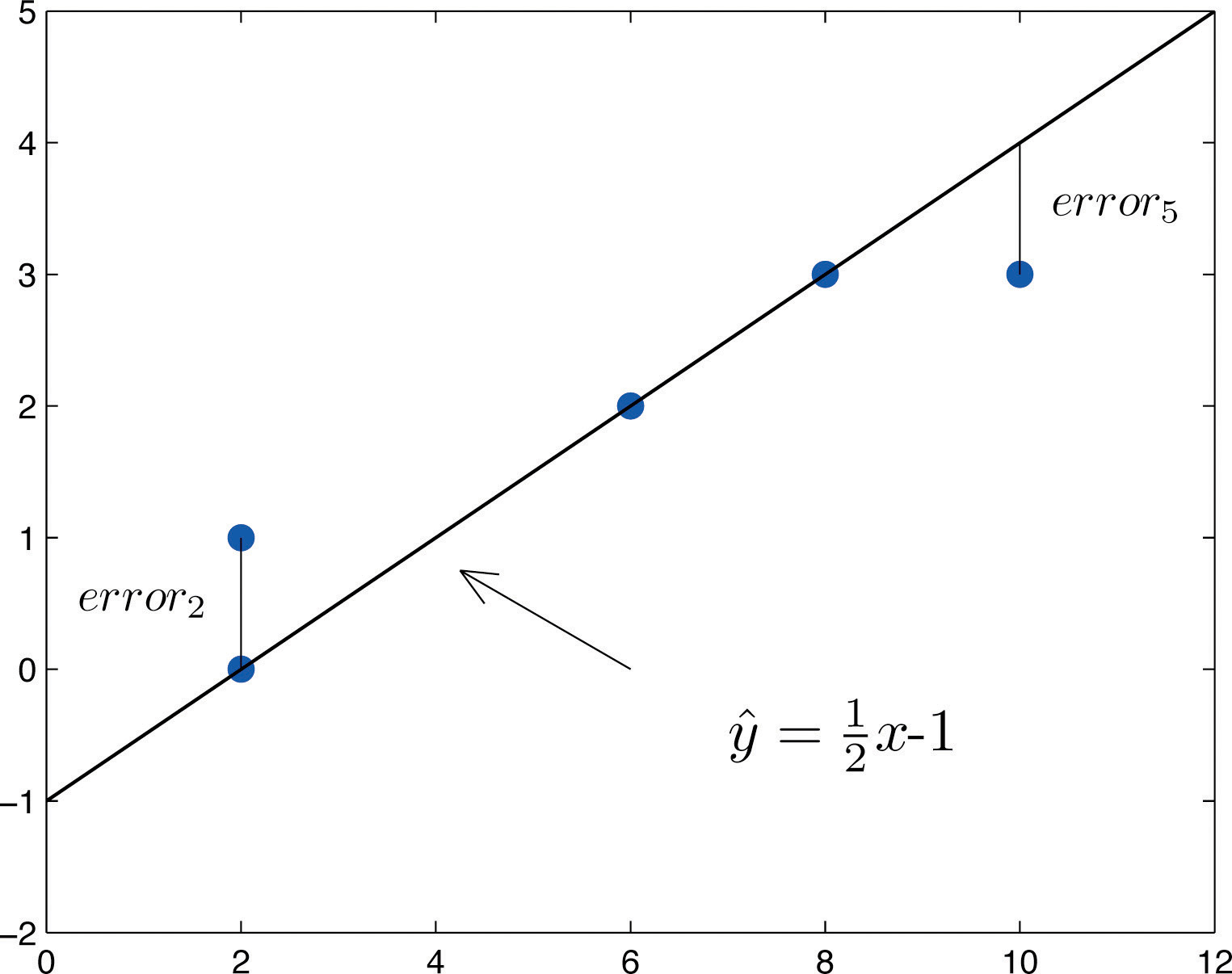
The calibration curve is fitted using regression analysis to fit different models to experimental data. A calibration curve is established to express the relationship between the response and the standard concentration for physical, chemical, and biological sensors. 
Calibration detects the response using a series of samples of the known concentration and sufficient purity. In terms of the user of the instrument, the structure of the instrumentation is so complex that it is difficult to adjust. Īccording to the definition of Dux, the calibration equation is used to verify the response of an instrument to the known properties of a material. Most quantitative analytical techniques for chemical analysis, such as spectrometry, Inductively Coupled Plasma Mass Spectrometry (ICP-MP) or electrophoresis, require a calibration curve to express the relationship between the response of the measuring technique and the standard concentration of the target analyst. The performance characteristics include accuracy, precision and sensibility of the sensors or instrument is so important, especially in chemical analysis. The method for this study can be used for other chemical instruments to establish an adequate calibration equation and ensure the best performance. No universe calibration equation could be found for these data sets. The adequate calibration equations with the data sets obtained with the same equipment and laboratory indicated that the adequate calibration equations differed. When outliers are removed, this calibration equation’s fit and prediction ability is significantly increased. The logarithmic transformation of the response is used to stabilize non-constant variance in the response data. Different forms of calibration equations are proposed. Nonlinear equations are suited to most of the data sets. The results of this study show that linear and higher order polynomial equations do not allow accurate calibration equations for many data sets. Suspected outliers in the data sets are checked. Residual plots are used as quantitative criteria. The Prediction Sum of Squares ( PRESS) statistic is used to compare the prediction ability. The standard error of the estimate errors, s, was used as criteria to determine the fitting performance. Suspected outliers in the data sets are verified. A constant variance test was performed to assess the suitability of calibration equations for this dataset. Four types of calibration equations are proposed: linear, higher-order polynomial, exponential rise to maximum and power equations. This study uses data sets from previous studies. Most studies use linear and polynomial equations. 
An adequate calibration equation ensures the performance of these instruments. 
The calibration equation verifies the response of a chemical instrument to the known properties of materials and is established using regression analysis. A calibration curve is used to express the relationship between the response of the measuring technique and the standard concentration of the target analyst. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
